 This additional energy is also been released as an electromagnetic wave which is able to carry a discrete amount of energy. Therefore, if an electron in a higher energy level needs to move downwards to an orbital with lower energy, it needs to release its additional energy. This is the inverse process of absorption, where energy is given out. The same explanation holds true for the case of emission as well. If enough energy is supplied by the energy source, the electrons might be able to absorb this energy and get excited to an extent where it leaves the atomic orbitals. However, the energy provided by the electromagnetic waves need to match the transitioning energy gap between the orbitals. Depending on the energy provided by the electromagnetic wave, the electron can either move up to the next energy level or to a higher one skipping several levels. Therefore, the process in which an electron accepts a discrete amount of energy (delivered to it by an electromagnetic wave) and elevates itself to a higher energy level is known as ‘absorption’. In addition, this transfer of energy takes place between the electron and the wave at an optimum state. It has been proven that electromagnetic waves also carry discrete amounts of energy rather than energy in a continuous form. For this to happen, the energy carried by the electromagnetic wave has to be equal to the energy gap between the orbitals. When electrons interact with electromagnetic radiation, they absorb its energy and are able to elevate into higher energy level orbitals within the atom. The electrons residing in these orbitals also carry the same amount of discrete energy. It means that this energy is not continuously variating and takes certain values. The energy of the orbitals around the nuclei of atoms are discrete. 
This is the main difference between absorption and emission. 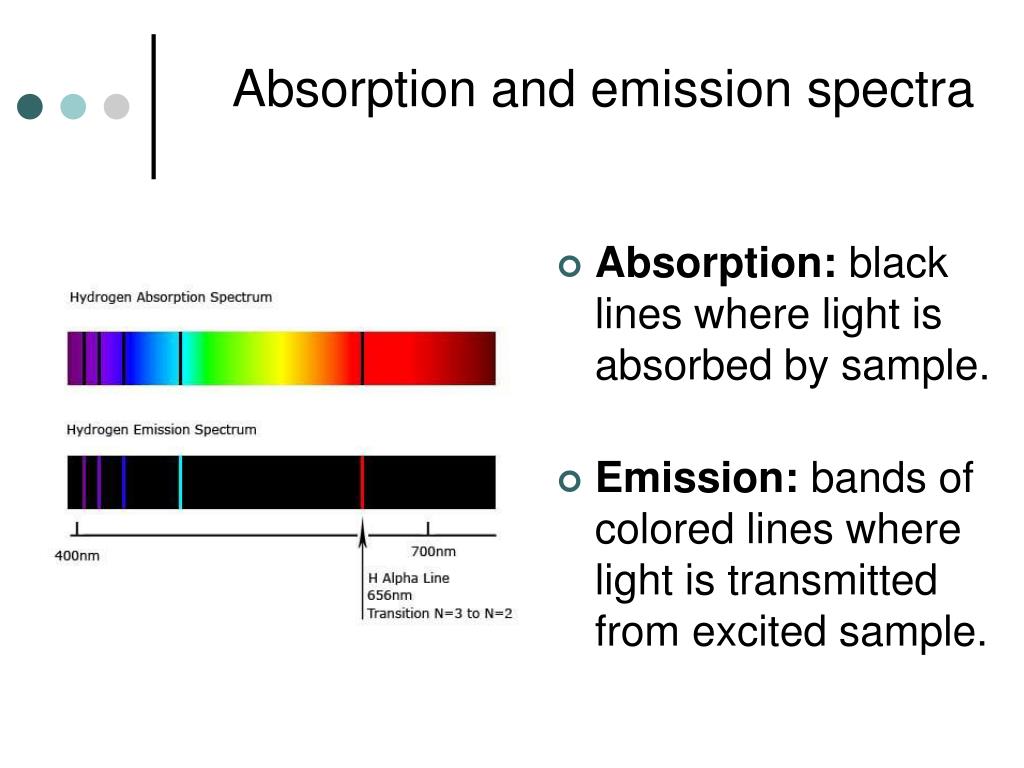
Therefore, an electron in a lower energy level has to absorb energy in order to move to a higher energy level and similarly an electron in a higher energy level has to emit an equivalent amount of energy in order to move down to a lower energy level. Consequently, the electrons residing in the lower energy levels carry lower energy and the ones occupying the higher energy levels carry a higher amount of energy respectively. The energy shells closer to the nucleus are less in energy and the energy increases as further it gets from the nucleus. Each atom is made up of a dense nucleus and a vast area of empty space which consists of energy shells where electrons reside. Absorption and emission are two common phenomena associated with electron transitions within energy levels of an atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
